Case Report
Volume 2 Issue 1 - 2020
Papilledema Due to Elevated Cerebrospinal Fluid Protein without Pleocytosis
Flaum Eye Institute 601 Elmwood Ave Box 659 Rochester, NY 14642
*Corresponding Author: Zoe R. Williams, Flaum Eye Institute 601 Elmwood Ave Box 659 Rochester, NY 14642.
Received: February 11, 2020; Published: February 19, 2020
Abstract
Purpose: To present the differential diagnosis and appropriate workup for patients with papilledema with elevated CSF total protein but no pleocytosis.
Case Report: Many patients are referred to Neuro-ophthalmology with a diagnosis of idiopathic intracranial hypertension who do not fulfill the diagnostic criteria. This case illustrates an example of vision-impairing papilledema with elevated CSF total protein without pleocytosis secondary to diabetes, severe hypothyroidism and multi-level spinal stenosis with disk herniation, which was rapidly corrected by optic nerve sheath fenestration (ONSF) with restoration of baseline vision. His disease course was complicated by subretinal hemorrhage from peripapillary choroidal neovascularization (PPCNV), which resolved with three intravitreal injections of bevacizumab.
Conclusion: Thorough workup of papilledema with elevated CSF total protein without pleocytosis can lead to unveiling of uncommon etiologies of papilledema. If causative etiologies cannot be corrected rapidly, ONSF remains an effective method to relieve papilledema and to preserve vision.
Keywords: Papilledema; Elevated CSF protein; Peripapillary choroidal neovascularization
Introduction
Papilledema occurs when increased intracranial pressure (ICP) disrupts axoplasmic flow within the optic nerve leading to axonal edema [1-4] and optic disc capillary leakage. It can be due to an intracranial mass, cerebral edema, changes in cerebrospinal fluid (CSF) production and absorption, venous outflow obstruction, obstructive hydrocephalus, or idiopathic intracranial hypertension (IIH). [4-6] Rarely, papilledema may also occur in the setting of an arteriovenous fistula, [4, 6, 7] chronic inflammatory demyelinating polyneuropathy (CIDP), [8] diabetic neuropathy, [8] thyroid dysfunction, [8] or from intraspinal lesions. [4, 8, 9] This case report highlights the appropriate workup of chronic papilledema in the setting of elevated CSF protein, complicated by development of peripapillary choroidal neovascularization (PPCNV), treated successfully with bilateral optic nerve sheath fenestration (ONSF) and intravitreal bevacizumab. Our patient had three potential etiologies for his elevated CSF protein: diabetes, severe hypothyroidism and multi-level spinal stenosis with disk herniation.
Case Report
A 45-year-old man with uncontrolled diabetes, hypertension, and obesity (BMI 43) presented with a two-month history of headache, binocular horizontal diplopia, and vision loss bilaterally. His best-corrected visual acuity at initial presentation was 20/70 (right eye) and 20/150 (left eye) with left afferent pupillary defect and reduced color vision on Hardy Rand and Rittler (HRR) plates (5/14 right eye and 1.5/14 left eye). His ocular exam was significant for bilateral disc edema with disc hemorrhages and peripapillary subretinal hemorrhages. Retinal exam also showed microaneurysms, exudate and intra-retinal hemorrhage consistent with diabetic retinopathy. Humphrey visual field testing showed an enlarged blind spot with visual field constriction in the right eye and diffuse depression in the left eye. He denied any use of minocycline, tetracycline, doxycycline, lithium, corticosteroids or excess vitamin A intake preceding his fulminant presentation with papilledema. MRI/MRV brain without and with gadolinium showed no mass lesion or venous sinus thrombosis but was suggestive of elevated ICP based on protrusion of the optic discs into the globes, expansion of the perioptic CSF space, and a partially empty sella. There was no abnormal T2 hyperintensity or enhancement of the optic nerves. Lumbar puncture (LP) showed an opening pressure of 34 cm H2O in right lateral decubitus position with CSF studies notable for high total protein (87), high glucose (188) and was otherwise normal (2 nucleated cells, negative Gram stain, and no growth on aerobic culture). He was started acetazolamide 500 mg extended-release BID with dose escalation to 1500 mg Qam and 1000 mg QHS.
Repeat LP showed opening pressure of 17.2 cm H2O on acetazolamide. CSF showed no nucleated cells, 1-2 red blood cells, glucose 190, total protein elevated at 167, negative Gram stain, aerobic culture, cytology and flow cytometry. He developed a subretinal hemorrhage with visual decline to 20/400 in his right eye due to PPCNV from elevated ICP, for which patient was treated with intravitreal bevacizumab. MRA brain was negative for dural AV fistula or central nervous system vasculitis. Exam by Neurology showed no evidence of acute or chronic inflammatory demyelinating polyneuropathy. MRI cervical/thoracic/lumbar spine was significant for multi-level spinal stenosis/disk herniation at C3-5, T8-10, and L4-L5, with a 6 mm right paramedian disc protrusion contacting the right descending L5 nerve root. Laboratory workup revealed elevated TSH (10.25), free T4 (0.7) and elevated thyroid peroxidase antibody (91). Serum protein electrophoresis to evaluate for possible POEMS syndrome showed no M protein.
Patient did not tolerate acetazolamide and self-discontinued therapy with worsening of his visual fields and papilledema prompting surgical intervention. He was treated with sequential ONSF with interval improvement in his visual acuity, color vision and visual field defects. His BCVA was 20/70 in both eyes. Color vision improved to HRR 11.5/14 in the right eye and 12.5/14 in the left eye. The subretinal hemorrhage resolved after three injections of bevacizumab. His papilledema improved significantly without signs of pallor.
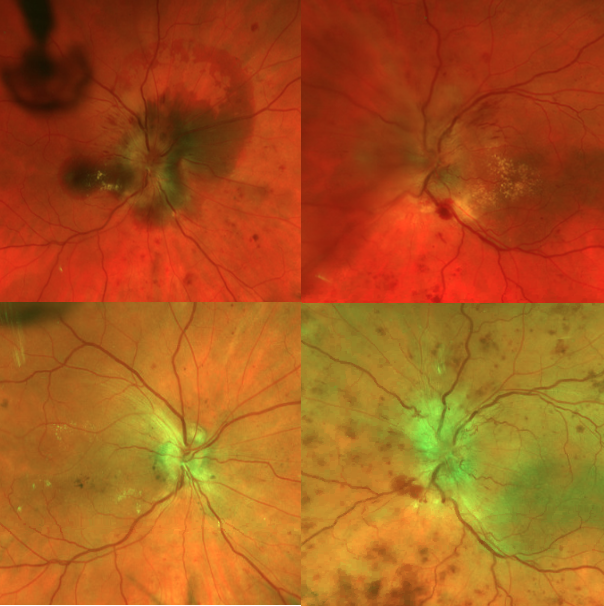
Figure 1: Top row shows florid papilledema in both eyes as well as subretinal hemorrhage in the right eye prior to treatment. Bottom row shows improved papilledema two weeks after ONSF and resolved subretinal hemorrhage after intravitreal bevacizumab treatment.

Figure 2: Top row shows patient’s Humphrey visual field prior to treatment. Bottom row shows repeat Humphrey visual field after ONSF and intravitreal bevacizumab.
Discussion
To summarize, our patient has three potential causes for his elevated CSF protein - diabetes, hypothyroidism and disc herniation. Diabetes has been reported to cause elevated CSF protein secondary to radicular demyelination [8] and appears to be duration-related. [10] Hypothyroidism has been shown to cause elevated CSF protein with the proposed mechanism of altered blood-brain barrier from low hormone levels rather than direct autoimmune response. [11] In fact, 6 out of the 15 patients with subclinical hypothyroidism had normal CSF protein despite the presence of thyroid antibodies. [11]
Large studies of patients with lumbar disc herniation have demonstrated elevated CSF protein presumed secondary to disrupted blood-nerve root barrier releasing plasma proteins into CSF. [12] On literature review, we found only one case report of disc herniation causing papilledema. [13] Michowiz et al. described a patient with disc herniation at T11-12 who had bilateral papilledema with normal CT and CTA head. [13] Interestingly, the opening pressure was normal and a lymphocytosis was identified with CSF total protein elevated at 150; however, there was resolution of his papilledema within 6 weeks of discectomy. [13]
The development of papilledema from spinal lesions is rare, especially after ruling out spinal tumors such as ependymomas. [4, 8, 13-15] In this case, it is plausible that our patient’s disc herniation caused mechanical stretching on the basement membrane along pia mater and subsequently disrupted the blood-nerve root barrier and lead to leakage of plasma protein into the CSF as suggested by Skouen et al. [12] Many authors have attributed elevated ICP to increased total protein blocking CSF reabsorption. [4, 5, 9, 13-15]
Our case adds to the current literature emphasizing the need to workup patients with papilledema and elevated CSF total protein without pleocytosis for underlying etiology of CSF protein elevation. The differential diagnosis includes spinal tumors, epidural abscess, epidural hematoma, intracranial hemorrhage, enlarging syrinx cavities, spondylitic myelopathy, CIPD, diabetic neuropathy, and thyroid dysfunction. [8-15] For patients like ours whose causative etiology cannot be rapidly corrected, localized surgical management with ONSF can be sight-saving.
Acknowledgement
This research was supported by an unrestricted grant from Research to Prevent Blindness
This research was supported by an unrestricted grant from Research to Prevent Blindness
References
- Tso, M. O., and S. S. Hayreh. (1977). “Optic Disc Edema in Raised Intracranial Pressure. IV. Axoplasmic Transport in Experimental Papilledema.” Archives of Ophthalmology (Chicago, Ill.: 1960) 95, no. 8: 1458–62.
- Sibony PA, Kupersmith MJ, Feldon SE, Wang JK, Garvin M. (2015). OCT Substudy Group for the NORDIC Idiopathic Intracranial Hypertension Treatment Trial. Retinal and Choroidal Folds in Papilledema. Invest Ophthalmol Vis Sci. 56(10): 5670–5680.
- Killer HE, Jaggi GP, Miller NR, et al. (2011). Cerebrospinal fluid dynamics between the basal cisterns and the subarachnoid space of the optic nerve in patients with papilloedema. British Journal of Ophthalmology 95: 822-827.
- Rigi M, Almarzouqi S, Morgan M, Lee A. (2015). Papilledema: epidemiology, etiology, and clinical management. Eye Brain. Aug 17 7: 47-57.
- van Crevel H. (1979). Papilloedema, CSF pressure, and CSF flow in cerebral tumours. J Neurol Neurosurg Psychiatry. 42(6): 493–500.
- Friedman, D. I, Liu, G. T, and Digre, K. B. ((2013)). “Revised Diagnostic Criteria for the Pseudotumor Cerebri Syndrome in Adults and Children.” Neurology. 81.13: 1159–1165.
- Biousse, V., and N. J. Newman. (2001). “Intracranial Vascular Abnormalities.” Ophthalmology Clinics of North America 14, no. 1: 243–64.
- Irani DN. (2009). Cerebrospinal Fluid in Clinical Practice, 1st edition. Philadelphia: Saunders Elsevier.
- Gardner, W. J., D. K. Spitler, and C. Whitten. (1954). “Increased Intracranial Pressure Caused by Increased Protein Content in the Cerebrospinal Fluid; an Explanation of Papilledema in Certain Cases of Small Intracranial and Intraspinal Tumors, and in the Guillain-Barre Syndrome.” The New England Journal of Medicine 250, no. 22: 932–36.
- Kobessho H, Oishi K, Hamaguchi H, Kanda F. (2008). Elevation of cerebrospinal fluid protein in patients with diabetes mellitus is associated with duration of diabetes. Eur Neurol 60: 132-36.
- Nyström E et al. (1997). Cerebrospinal fluid proteins in subclinical and overt hypothyroidism. Acta Neurol Scand. 95(5): 311-4.
- Skouen JS, Larsen JL, Gjerde IO, Hegrestad SE, Vollset SE. (1997). Cerebrospinal fluid protein concentrations in patients with sciatica caused by lumbar disc herniation: an investigation of biochemical, neurologic, and radiologic predictors of long-term outcome. J Spinal Disord. 10(6): 505-11.
- Michowiz, S. D., H. Z. Rappaport, I. Shaked, A. Yellin, and A. Sahar. (1984). “Thoracic Disc Herniation Associated with Papilledema. Case Report.” Journal of Neurosurgery 61, no. 6: 1132–34.
- Teng, P., J. H. Wagner, and M. W. Buxbaum. (1960). “Giant Ependymoma of the Spinal Cord Associated with Papilledema. Review of Literature and Report of a Case.” Archives of Neurology 2: 657–62.
- Schijman, E., G. Zúccaro, and J. A. Monges. (1981). “Spinal Tumors and Hydrocephalus.” Child’s Brain 8, no. 6: 401–5.
Citation: Xu He MD, Zoë R. Williams MD. (2020). Papilledema Due to Elevated Cerebrospinal Fluid Protein without Pleocytosis. Journal of Ophthalmology and Vision Research 2(1). DOI: 10.5281/zenodo.3722852
Copyright: © 2020 Zoë R. Williams. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
