Case Report
Volume 2 Issue 1 - 2020
Glaucoma Drainage Device (GDD) Implantation Following Ultrasound Cycloplasty (UCP) in Aphakic Glaucoma
Ophthalmology Department, Hospital Universitario HM Madrid, Madrid, Spain
*Corresponding Author: Teresa Colás-Tomás MD, Pl. del Conde del Valle de Súchil, 16, 28015 Madrid, Spain.
Received: November 11, 2020; Published: November 23, 2020
Abstract
Purpose: A case of an aphakic glaucoma treated with two ultrasound circular cyclocoagulation procedures and posterior implantation of Ahmed valve device is presented.
Patients and Methods: Forty-year-old female with aphakic glaucoma in both eyes and elevated intraocular pressure in her single functioning right eye. Since this case involved a low vision eye and special anatomical features, it was decided to perform localized Ultrasound Circular Cyclo-Coagulation (UC3®)
Results: At 2 months it was necessary to repeat the procedure with UC3. At 6 months it was decided to implant an Ahmed valve device due to poor intraocular pressure control. The scleral tunnel was created 3 mm away from the corneoscleral junction and in between two scleral thinning marks, so as to minimize the risk of complications. At 3 years of follow-up, visual acuity remains stable and IOP is 14 mmHg.
Conclusions: UC3 can be indicated for eyes that have not undergone surgery and where, due to their anatomical and functional characteristics, it is advisable to use a fast and safe technique that ensures a prompt recovery. Several consecutive ultrasound cycloablation procedures do not preclude the possibility of a subsequent Ahmed-valve implantation, as long as the previously-treated scleral areas are avoided.
Keywords: Aphakic glaucoma; Ultrasound circular cyclocoagulation; Ultrasound cycloablation; HIFU; UC3
Abbreviations: UC3®: Ultrasound Circular Cyclo-Coagulation; OS: Left eye; OD: Right eye; VA: Corrected visual acuity; IOP: intraocular pressure; OCT: optical coherence tomography; HIFU: high-intensity-focused ultrasound
Introduction
Aphakic glaucoma is one of the most relevant and common complications occurring after congenital-cataract surgery. It is a complex type of glaucoma, which often requires the application of different surgical techniques to have it under control [1-3]. Trabeculectomy with Mitomycin C or glaucoma drainage device (GDD) implantation have typically been the first-line surgical options, while traditional cyclo-ablative procedures where kept for refractory cases only, due to the multiple and severe complications that were associated with this approach.
However, the recent advent of new "soft" cyclo-destructive or cyclo-modulating techniques that make procedures safer, more repeatable and with favorable outcomes, has made it possible to include cyclo-ablation within the first therapeutic step. Moreover, it is worth bearing in mind that these techniques do not prevent other more aggressive surgeries from being performed at a later stage.
Clinical Case
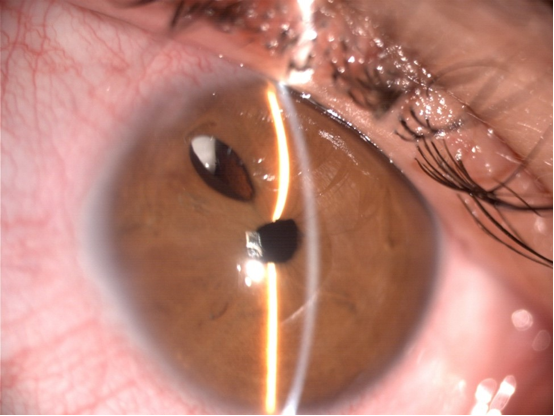
A 40-year-old female with aphakia (following bilateral congenital-cataract surgery at age 5) and aphakic glaucoma in both eyes, who has been under follow-up for the past 14 years. In her left eye (OS) she had a retinal detachment with a poor clinical course. Her best-corrected visual acuity (VA) in her right eye (OD) is 0.1—her correction being +5 D (sphere) and -6.50 D at 135º (cylinder)—while with her OS she does not perceive any light. The slit-lamp examination in her OD showed horizontal nystagmus, conjunctival hyperemia, surgical aphakia with a 10-o’clock iridectomy and the presence of retropupillary cortical remains (Figure 1). The gonioscopy exam revealed angle dysgenesis with discontinuous peripheral anterior synechiae. Central corneal thickness measured with ultrasound pachymetry was 514 µm, while intraocular pressure (IOP) measured with Goldman applanation tonometry amounted to 36 mmHg, despite it being treated with 4 different drugs. Eye fundus examination, which is performed with significant difficulty, showed a pale optic disc with a 0.9 cup-to-disc ratio. Quality perimetry and tomography could not be performed due to the patient's low VA and the presence of nystagmus.
Since this case involved a single functioning eye that had with low vision, special anatomical features and poor IOP control, it was decided to perform localized Ultrasound Circular Cyclo-Coagulation (UC3® [Eye Tech Care, Rillieux La Pape, France]) so as to decrease aqueous humor production and to minimize potential surgery-related complications.
In order to apply a correct and properly-focused treatment upon the ciliary body, an optical coherence tomography image (OCT-Visante platform) was taken prior to surgery; this allowed us to calculate the optimum size of the probe to be later used.
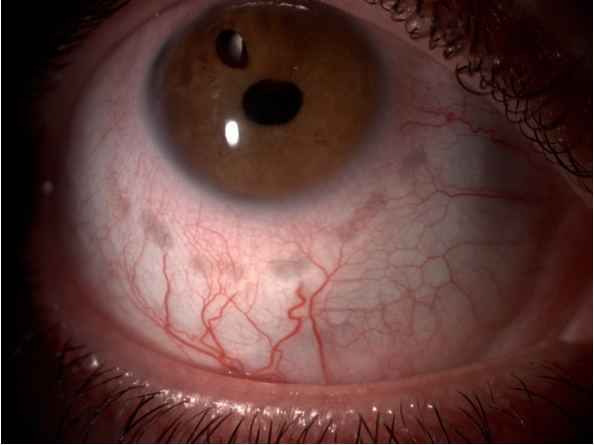
The patient underwent sub-Tenon's anesthesia and sedation before the procedure, which was then performed with a 13 mm probe and the following parameters: frequency= 20.636 MHz; duration= 4 s; number of activated sectors= 6; acoustic power= 0.7 W and time between consecutive shots= 20 s. As a result of the procedure IOP dropped to 9 mmHg at 2 weeks, but it went back up to 23 mmHg at 2 months with two IOP-lowering drugs being needed. Scleral burns were observed in the area where the probe had been applied (Figure 2).
Due to the simplicity of the initial treatment and the partial IOP reduction achieved within the first weeks, it was decided to repeat the procedure (with the same parameters) in the six contiguous sectors. Hence, IOP was brought down to 7 mmHg a month after surgery, with a subsequent gradual increase to 24 mmHg 4 months later, despite topical treatment with 3 drugs.
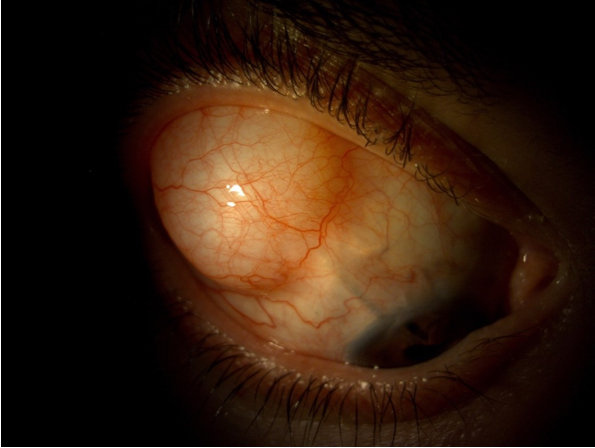
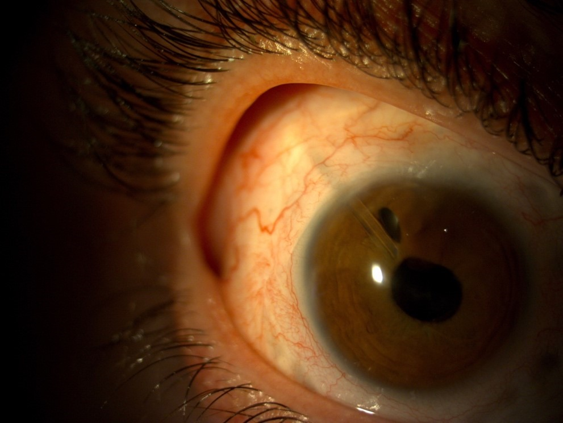
Given the patient's poor intraocular pressure control and highly compromised low VA, it was decided to implant an Ahmed valve device® (AGV; New World Medical, Inc., Rancho Cucamonga, CA, US) in the upper temporal quadrant. The scleral tunnel was created 3 mm away from the corneoscleral junction and in between two scleral thinning marks, so as to minimize the risk of extrusion, scleral dellen or infection (Figure 3). Mitomycin C was not employed in this procedure but, instead, an autologous scleral graft together with Ologen® (Aeon Astron Corporation, Taipei, Taiwan) were used. The tube was placed in the anterior chamber; more specifically, in the vicinity of the iridectomy (Figure 4). From the very first moment the evolution was positive, with the patient maintaining her VA and reaching an IOP of 9 mmHg at 1 week, 14 mmHg at 2 months ,16 mmHg at 1 year and 14 mmHg at 3 years of follow-up with concurrent use of one drug.
Discussion
Ultrasound circular cyclo-coagulation (currently called ultrasound cycloplasty) is an emerging technique that relies on HIFU (high-intensity-focused ultrasound) technology to, in an automated and reproducible manner, decrease aqueous humor production through thermal coagulation of the ciliary body’s epithelium [14,19]. It acts precisely and selectively upon the epithelium, thus preventing damage to adjacent tissues. Some studies suggest that it can also stimulate aqueous humor drainage (outflow) through the uveoscleral pathway [12,13]. Ultrasound circular cyclo-coagulation (i.e., ultrasound cycloplasty) is considered a safer and less invasive technique than the traditional surgical approaches, thus enabling a faster post-surgical recovery.
Its use has been widely assessed in glaucoma cases that are refractory to other surgical treatments [4-7]. Following the good outcomes and the few complications seen with this technique, several studies have recently been published focusing on naïve eyes that had not previously undergone surgery [8-10]. Their resulting IOP decrease ranges from 26% to 42%, with hypotensive efficacy being maintained after 6 or 12 months (depending on the particular study) of follow-up [4-10, 15,16]. Moreover, there does not seem to be any differences—in terms of IOP decrease—between naive eyes and eyes that had previously undergone surgery [18,19].
However, little experience has been gathered to date in aphakic glaucoma cases. In Chamard et al’s study, which included 100 patients, there were 4 cases of aphakic glaucoma and UC3 yielded successful outcomes for all of them [10]. A greater hypotensive efficacy has been described for angle-closure glaucoma cases [7,18].
The possibility of retreating a given eye several times with this technique makes it a very attractive alternative in certain patient cases. De Gregorio et al demonstrated the safety, and even a greater hypotensive efficacy, in patients receiving up to 3 UC3 treatments at 4-month intervals [17]. According to the data published in the literature, re-treatment rates differed significantly, depending on the specific study: 6.2% at 6 months, 1.1% at 6 months, 29.4% at 6 months and 50% at 4 months [10, 8, 6, 17]. In our particular case, re-treatment was required at 4 months, and then a more aggressive surgery had to be considered due to insufficient IOP control being achieved.
Due to the scleral thinning that occurs after UC3 and the lack of literature on the subject, we did not find it appropriate to use techniques involving a scleral flap; for this reason, we considered GDD implantation to be a safer method, with the scleral tube being placed in between two scleral marks. The good evolution and long-term outcomes lead us to think that UC3 can be a safe alternative in aphakic glaucoma cases, although more subjects and longer follow-up periods would be necessary to achieve a comprehensive assessment of this approach.
Conclusion
UC3 can be indicated for eyes that have not undergone surgery and where, due to their anatomical and functional characteristics, it is advisable to use a fast and safe technique that ensures a prompt recovery.
Several consecutive ultrasound cycloablation procedures do not preclude the possibility of a subsequent Ahmed-valve implantation, as long as the previously-treated scleral areas are avoided.
Conflict of interest statement
The authors not have proprietary interest
The authors not have proprietary interest
Patient consent confirmation
The patient consent to publish her case and images.
The patient consent to publish her case and images.
References
- Zetterberg M, Nyström A, Kalaboukhova L, Magnusson G. (2015). Outcome of surgical treatment of primary and secondary glaucoma in young children. Acta Ophthalmol 93: 269-275.
- Bhola R, Keech RV, Olson RJ, Peterson DB. (2006). Long term outcome of pediatric aphakic glaucoma. J AAPOS 10: 243-248.
- Baris M, Biler ED, Guven Yilmaz S, Ates H, Uretmen O, Kose S. (2019). Treatment results in aphakic patients with glaucoma following congenital cataract surgery. Int Ophthalmol 39: 11-19.
- Aptel F, Dupuy C, Rouland JF. (2014). Treatment of refractory open-angle glaucoma using ultrasonic circular cyclocoagulation: a prospective case series. Curr Med Res Opin 30(8): 1599-605.
- Melamed S, Goldenfeld M, Cotlear D, Skaat A, Moroz I, (2015). High- intensity focused ultrasound treatment in refractory glaucoma patients : results at 1 year of prospective clinical study. Eur J Ophthalmol 25 (6): 483-9
- Denis P, Aptel F, Rouland J-F, Nordmann JP, Lachkar Y, Renard JP, et al. (2015). Cyclocoagulation of the ciliary bodies by high- intensity focused ultrasound: a 12-month multicenter study. Invest Ophthalmol Vis Sci 56 (2): 1089-96.
- Giannaccare G, Vagge A, Gizzi C, Bagnis A, Sebastiani S, Del Noce C, et al. (2017). High-intensity focused ultrasound treatment in patients with refractory glaucoma. Graefes Arch Clin Exp Ophthalmol 255 (3): 599-605.
- Aptel F, Denis P, Rouland JF, Renard JP, Bron A. (2016). Multicenter clinical trial of high-intensity focused ultrasound treatment in glaucoma patients without previous filtering surgery. Acta Ophthalmol 94(5): e268-77.
- Deb-Joardar N, Reddy KP. (2018). Application of high intensity focused ultrasound for treatment of open-angle glaucoma in Indian patients. Indian J Ophthalmol 66(4): 517-523.
- Chamard C, Daien V, Han H, Green field DS, Villain M. (2020). Efficacy and safety of ultrasound circular cyclocoagulation with second-generation probe in glaucoma: a retrospective study. PLoS One 15 (1): e02273389.
- Aptel F, Charrel T, Palazzi X, Chapelon JY, Denis P, Lafon C. (2010). Histologic effects of a new device for high-intensity focused ultrasound cyclocoagulation. Invest Ophthalmol Vis Sci 51: 5092-5098.
- Mastropasqua R, Agnifili L, Fasanella V, Toto L, Brescia L, Di Staso S, et al. (2016). Uveo-scleral outflow pathways after ultrasonic cyclocoagulation in refractory glaucoma: an anterior segment optical coherence tomography and in vivo confocal study. Br J Opthalmol 100: 168-1675.
- Aptel F, Begle A, Razavi A, et al. (2014). Short and long term effects of the ciliary body and the aqueous outflow pathways of high-intensity focused ultrasound cyclocoagulation. Ultrasound Med Biol 40: 2096-106.
- Mastropasqua R, Fasanella V, Mastropasqua A, Ciancaglini M, Agnifili L. (2017). High-intensity foused ultrasound circular cyclocoagulation in glaucoma: a step forward for cyclodestuction? Journal of Ophthalmology 7136275.
- Hugo J, Matonti F, Beylerina M, Zanin E, Aptel F, Denis D. (2019). Safety and efficacy of high-intensity focused utrasound in severe or refractory glaucoma. Eur J Ophthalmol 25: 1120672119874594.
- De Gregorio A, Pedrotti E, Stevan G, Montali M, Morselli S. (2017). Safety and efficacy of multiple cyclocoagulation of ciliary bodies by high-intensity focused ultrasound in patients with glaucoma. Graefes Arch Clin Exp Ophthalmol 255(12): 2429-2435.
- Hu D, Tu S, Zuo C, Ge J. (2018). Short – term observation of ultrasonic cyclocoagulation in chinese patients with end stage refractory glaucoma: a restrospective study. J Ophthalmol; 2018: 4950318.
- Posarelli C, Covello G, Bendinelli A, Fogagnolo P, Nardi M, Figus M. (2019). High-intensity focused ultrasound procedure: The rise of a new noninvasive glaucoma procedure and its possible future applications. Surv Ophthalmol. 6: 826-834.
Citation: Aitor Fernández García MD and Teresa Colás-Tomás MD. (2020). Glaucoma Drainage Device (GDD) Implantation Following Ultrasound Cycloplasty (UCP) in Aphakic Glaucoma. Journal of Ophthalmology and Vision Research 2(1). DOI: 10.5281/zenodo.4290075
Copyright: © 2020 Teresa Colás-Tomás. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.